CureDuchenne Congratulates Myosana on Closing its Latest Round of Funding

Myosana Therapeutics, a biotech company based in Seattle, WA, closed a new $5 million financing round to support further development of their non-viral gene therapy technology for the treatment of Duchenne muscular dystrophy and other neuromuscular diseases. Their non-viral delivery platform is aimed at overcoming the limitations of current AAV-delivered gene therapies, to potentially deliver the full-length dystrophin gene and allow for re-dosing of patients.
CureDuchenne Ventures identified the potential of this approach years ago, and was the first investor in Myosana, in April 2020. This new financing is not just validation in terms of Myosana’s progress, but also of CureDuchenne’s venture philanthropy model. Part of CureDuchenne Venture’s mission is to be a catalyst, accelerating the pace of research and helping companies gain the traction they need to attract new investors. To that end, we welcome PPMD and MDA to join us in support of Myosana’s research, as developing truly transformative therapies requires the attention and focus of all of us in the Duchenne community.
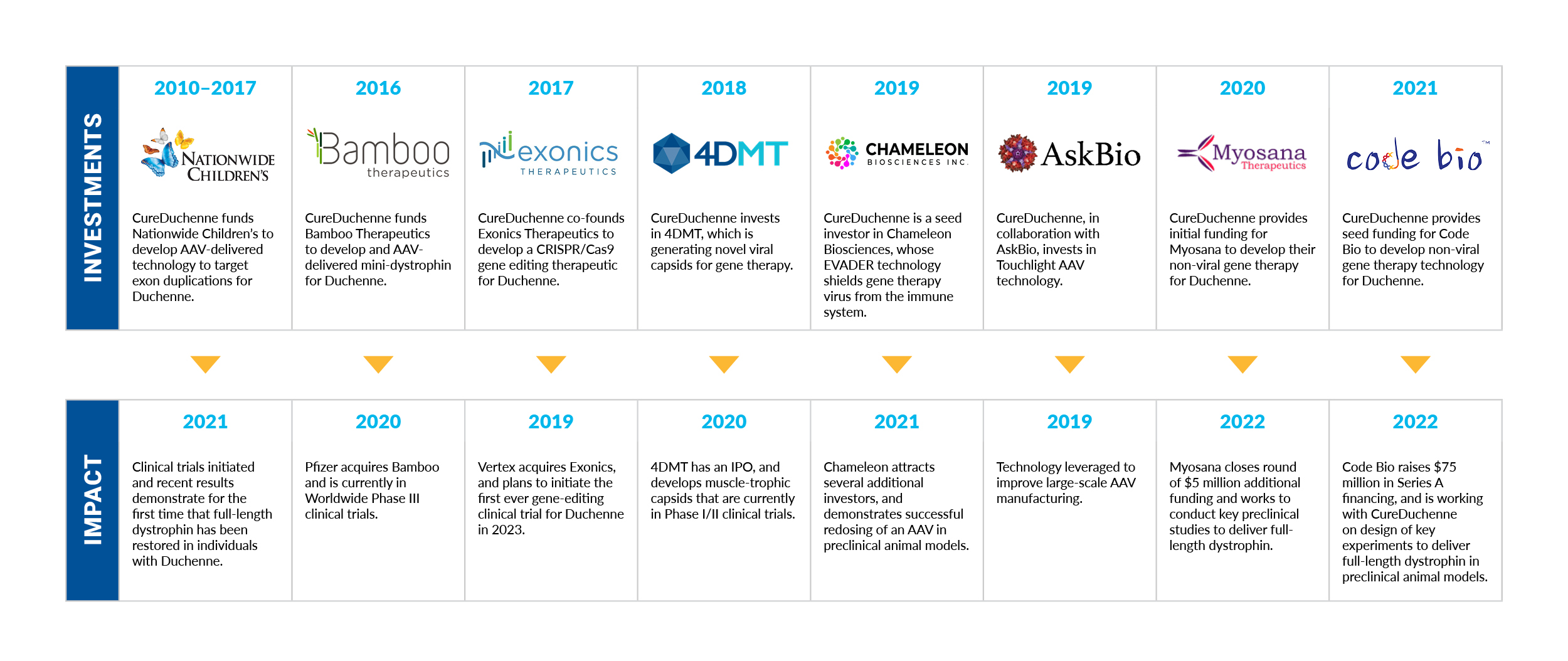
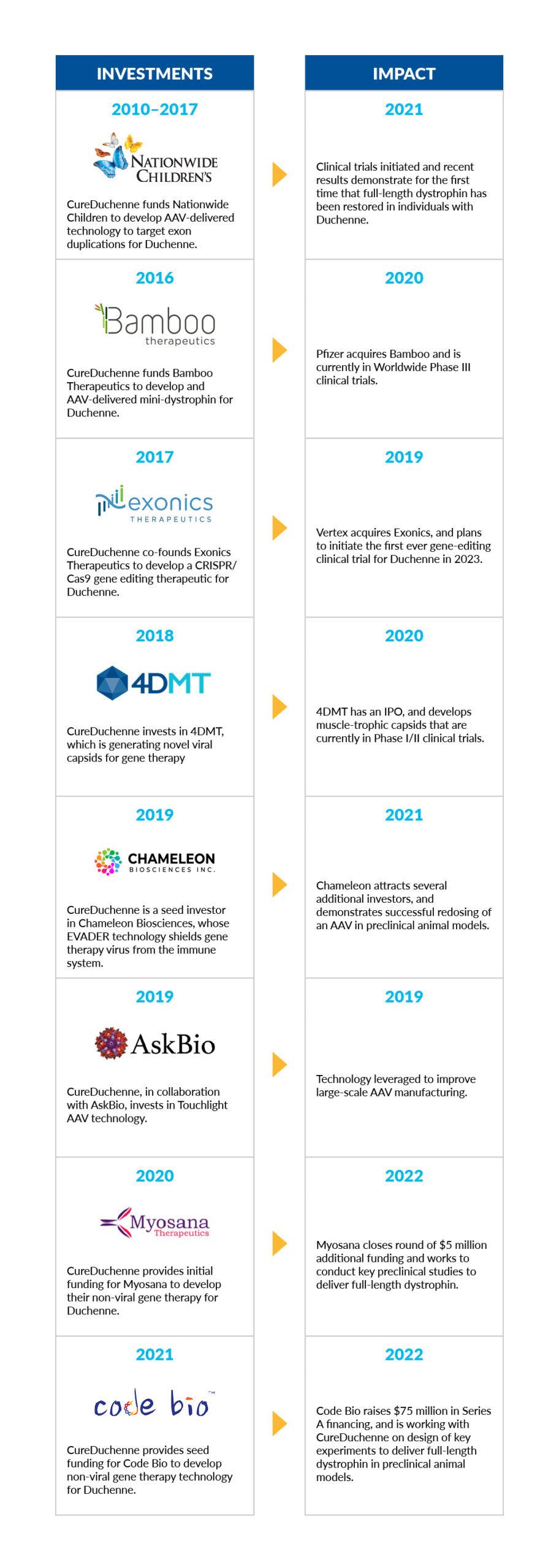
CureDuchenne has a long history of supporting gene therapy and gene editing research in Duchenne, as demonstrated in the table, below. Overcoming the current limitations of AAV-gene therapy approaches will continue to be a focus in the coming years.
CureDuchenne Investment Timeline
CureDuchenne would like to thank those in the Duchenne community who helped us raise the money used for this initial seed investment to advance Myosana’s early science to where it is today. CureDuchenne has long believed in the promise of gene therapy, and has made several key investments to facilitate gene therapy and gene editing programs to treat Duchenne.
Myosana’s news today is further proof that our venture philanthropy model is continuing to work on behalf of everyone in the Duchenne community to accelerate science and deliver better treatments faster.
Read more information about our initial investment to Myosana and the science here.